Polybalm® study: Nail damage caused by chemotherapy
Share
The Polybalm® Study: A double-blind, randomized study of a polyphenol-rich nail bed balm for chemotherapy-induced onycholysis (UK Polybalm Study)
The Polybalm® Study. A double-blind, randomized study of a polyphenol-rich nail bed balm for chemotherapy-induced onycholysis: the UK Polybalm Study
Thomas, R., Williams, M., Cauchi, M. et al. A double-blind, randomized trial of a polyphenol-rich nail bed balm for chemotherapy-induced onycholysis: the UK Polybalm Study. Breast Cancer Res Treat 171, 103–110 (2018). https://doi.org/10.1007/s10549-018-4788-
Abstract
Purpose
Nail damage is common in patients undergoing chemotherapy and leads to disfigurement and pain. This study evaluated whether a topical balm containing steam-extracted, bioactive, polyphenol-rich herbal oils blended with organic waxes could protect the nails based on its reported anti-inflammatory, analgesic, antioxidant, and antimicrobial properties.
methods
Sixty patients (23 men, 37 women) were randomized to apply either the plant balm (PB) or a petrolatum-based control substance (PC) to their nail beds two to three times daily. Demographics, type, and number of chemotherapy cycles did not differ between the two groups; recruitment took place between September 2015 and September 2016. An unpaired t-test was used to assess differences in symptoms and physical nail damage between the two groups.
Results
Symptom scores, as measured by the Dermatology Life Quality Questionnaire (DLQQ), were significantly better in the PB group compared to the PC group between the start and end of chemotherapy. Similarly, the mean reduction in nail damage, assessed by the treating physician using the Nail Psoriasis Index, was also significantly different.
conclusion
The polyphenol-rich essential oils and plant-based waxes in this nail bed balm significantly reduced chemotherapy-related nail damage and improved nail-related quality of life compared to a control substance. A further analysis is planned combining this balm with nail bed cooling.
Discussion and implications for clinical practice
Scientific evidence for practical strategies to reduce this distressing and cosmetically noticeable side effect of one of the most commonly used chemotherapy drugs has long been overdue. To date, many patient organizations have recommended using dark nail polish or standard petrolatum-based nail balms to protect nails during chemotherapy; however, there is little published evidence of their effectiveness or even anecdotal reports of benefit [1]. Cooling the nails during chemotherapy can reduce nail toxicity, but commercially available gloves are rarely used in the UK due to practical concerns among healthcare workers regarding limited access to patients' hands . The significantly lower levels of physical nail damage and improvements in nail-related quality of life in the cohort using the plant-based balm in this robustly designed RCT strongly suggest that this balm will benefit future patients in routine clinical practice. Since nail toxicity continued to occur in a minority of participants who also had pronounced general side effects from chemotherapy, further research combining this balm with cooling would be warranted.
Image 1
Introduction
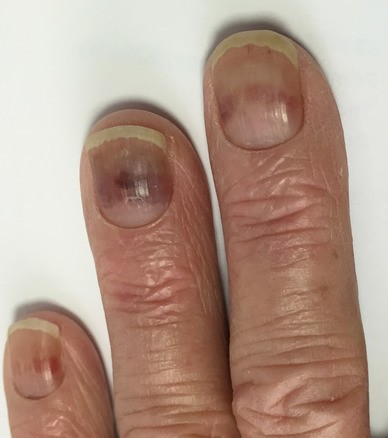
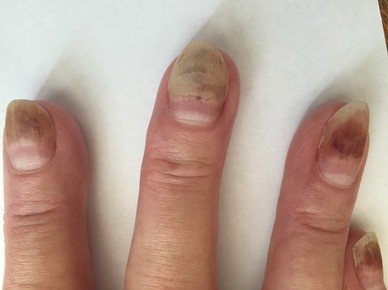
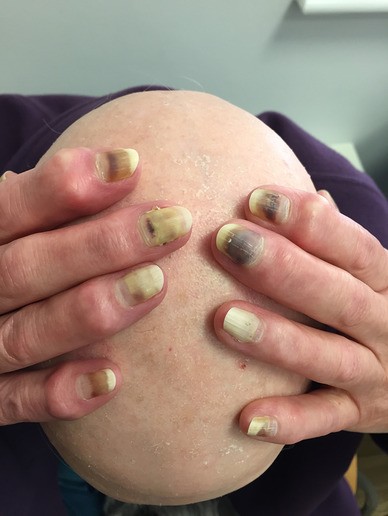
Chemotherapy-induced onycholysis is caused by disorganized inhibition of rapidly dividing cells in the nail bed [2,3,4,5]. Other contributing factors are related to the anti-angiogenic properties of taxanes and excessive local inflammatory processes [2,3,4,5]. Disruption of normal anatomy allows bacterial and fungal pathogens to invade the nail beds, causing additional damage [2,5]. In most cases, this manifests as ridges in the nails corresponding to the timing of chemotherapy episodes, known as Beau's lines (Figure 1). More severe nail damage, especially in association with taxanes, can lead to discoloration, painful subungual bleeding (Figure 2), deformities and complete onycholysis, which is distressing, cosmetically noticeable, affects body image (Figures 3, 4), restricts activities of daily living and can lead to more serious consequences including secondary infections – the latter being particularly relevant in patients at risk for neutropenia [2,3,4,5,6].
Figure 1
Mild, asymptomatic nail changes corresponding to chemotherapy cycles — Beau lines
Figure 2
Unpleasant, chemotherapy-induced subungual bleeding
Figure 3
Painful chemotherapy-induced partial onycholysis
Figure 4
Severe, debilitating subungual bleeding and secondary infection with complete onycholysis
Cooling the fingers has been described as a method to reduce docetaxel-induced nail toxicity by inducing vasoconstriction and slowing onychocyte metabolism [7, 8, 9]. However, this technique has not been widely adopted in the United Kingdom due to concerns among oncology nurses regarding limited access to patients' hands . Patient groups recommend various anecdotal strategies, including applying nail polish and massaging petrolatum-based moisturizing balms into the nail bed, but there have been no robust prospective studies to support these practices [1].
The plant-based waxes and essential oils selected for this study are naturally rich in phytochemicals, particularly from the group of phenolic polyphenols, which are attributed with antioxidant, anti-inflammatory, DNA repair-promoting, and antimicrobial properties [10–17]. In addition to their ability to moisturize the skin and thus prevent dryness and splitting of the nails, it has been hypothesized that they are sufficiently absorbed into the nail beds to act as a local antidote to chemotherapy, preventing damage to proliferating stem cells and avoiding secondary damage from inflammation or secondary infection [10–19].
methodology
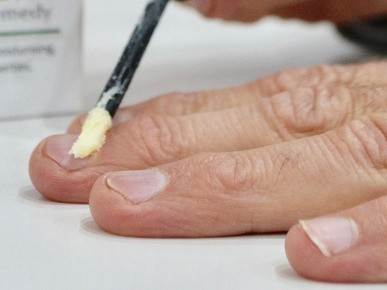
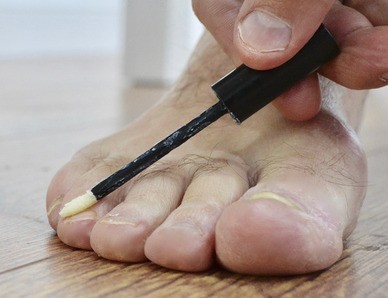
The study recruited over 13 months at Bedford Hospital, one of the hospitals affiliated with the University of Cambridge. A total of 23 men with prostate cancer and 37 women with breast cancer receiving chemotherapy were randomized between September 2015 and October 2016, out of a total of 63 patients considered for this study. One person declined after reading the information materials, and two had pre-existing nail conditions [see CONSORT diagram]. Following written informed consent, randomization took place when the attending physician contacted the study office, which then opened the next sealed envelope in strict numerical order. The randomized groups were demographically balanced with respect to the number of chemotherapy cycles received, sex, type of chemotherapy, baseline nail condition, and age (see Table 1). The balms were supplied in 50 ml jars, labeled A or B, along with an applicator. They were similar in color, consistency, and odor. Participants were instructed to apply the assigned balm to their fingernail beds 2–3 times daily until the end of chemotherapy (Figures 5, 6). Neither members of the research team, physicians , participants, nor the statisticians knew the contents of the respective jars until the final results had been analyzed.
Table 1 Demographics
Figure 5: Polybalm applied to the cuticles, surrounding skin and nail beds of the fingers to maintain nail health.
Figure 6: Polybalm applied to the cuticles of the toenails
At the start and end of the study, patients recorded symptoms related to their nail health using the validated Dermatology Life Quality Questionnaire and a Linear Severity Scale (LSS). Physicians documented the physical appearance of the nails using the validated Nail Psoriasis Severity Index (NPSI) and the Linear Severity Scale. Photographs of the nails were also sent to three other independent physicians for external validation.
Ingredients of the balms
The placebo balm consisted of a blend of unrefined shea butter (Butyrospermum parkii), organic beeswax (Cera alba), extra virgin organic cold-pressed olive oil (Olea europaea), organic cocoa butter (Theobroma cacao), African sage (Tarchonanthus camphoratus), wintergreen leaf (Gaultheria procumbens), lavender flower (Lavandula spica), and eucalyptus leaf (Eucalyptus globulus). The oils were extracted from the plants by steam distillation and then gently blended, avoiding overheating to prevent damage to the phytochemicals. It contained no preservatives, parabens, sulfates, or perfumes. The placebo control balm contained hydrogenated petrolatum, candelilla wax, and lavender and eucalyptus essences to ensure a placebo effect.
Quality assurance
The study was designed with the support of the independent National Cancer Research Clinical Trials Development Committee, oncologists from Addenbrooke's Hospital, consultants from the Department of Biological Science at Coventry University, and an independent statistician. The study was externally audited to meet Good Clinical Practice (GCP) guidelines and the standards of the East of England—Cambridge Central Research Ethics Committee (REC reference: 15/EE/0357). The investigational medicinal products were manufactured specifically for this study by Power Health Ltd, Yorkshire. Additional stability and microbiological tests were carried out by Advantis Laboratories Ltd to comply with EU cosmetic standards (REC reference: 76/768/EEC).
Statistical considerations
The sample size was determined taking into account a previously reported incidence of chemotherapy-induced onycholysis of 40–70% [2–5]. The trial development committee considered a 20% worsening of the nail toxicity score to be clinically relevant. For power calculation, the nearest relevant study investigating the benefit of a frozen glove on the incidence of onycholysis was used , which included 20 or 25 patients, respectively, to achieve a statistical power of 80% for a two-sided test at α = 0.05. A Matlab 2011a calculation was used to determine the sample size. The resulting curve estimated that 50 patients would be sufficient to detect a 20% difference in toxicity with a √2 standard deviation (SD) for a two-sided test at α = 0.05 and 95% significance. The study committee therefore opted for 60 participants to account for unforeseen events or dropouts. The analysis compared the difference in nail damage scores between the start and end of chemotherapy, as well as between the herbal balm and control groups, using an unpaired t-test for all four measures of nail health at a significance level of α = 0.05 at 95%.
Results
Patient and physician measurements of nail toxicity showed large and statistically significant differences between the plant balm (PB) and placebo control (PC) groups (Table 2). In the PB group, the DLQQ score decreased by an average of 0.034 from baseline to the end of chemotherapy. In the PC group, the DLQQ score decreased by an average of 6.10. This resulted in a mean difference in the extent of decline in quality of life of 6.06, confidence intervals (CI) 4.17–7.95, significance p < 0.00001 in the unpaired t-test. For the Linear Severity Scale (LSS), the mean nail condition, as measured by the patients themselves, improved by 2.63 mm during chemotherapy in the PB cohort compared to a deterioration of 64.1 mm in the PC group (difference 66.72 mm; CI 52.97–80.47, p < 0.00001). Differences in the physical appearance of the nails were similarly large and statistically significant, as measured by the validated NAPSI and a standard linear analog scale. The values of the other nail health measures are summarized in Table 2.
Table 2 Summary of the average change in scores at the beginning and end of the study
It was interesting to observe that the two patients in the herbal balm group who nevertheless exhibited moderate damage also suffered from other serious chemotherapy complications, including neutropenic sepsis, diarrhea, and peripheral neuropathy. No balm-related adverse events were reported; however, one person in the herbal balm group discontinued use before the end of the first cycle, citing lack of time as the reason.
discussion
The polyphenol-rich essential oils and plant-based waxes in this nail bed balm significantly reduced the physical appearance and burden of chemotherapy-induced nail damage compared to a petrolatum-based control balm. Regarding the study design, an intra-individual comparison, applying one cream to one hand and the other to the opposite hand, might have seemed more relevant. However, this was rejected by the design committee as impractical because compliance violations would have outweighed any potential statistical benefits: participants would very likely have confused their assigned hands once differences became apparent. The second design question concerned the choice of placebo control. Since no published nail bed balm data were available at the start of the study, a statistically valid comparison would have been a test substance versus no substance. However, this would not have blinded the study; therefore, a simple, commonly used petrolatum-based balm product, to which essences were added by the manufacturers, was chosen. It was assumed that the control could bring some benefit through their moisturizing properties, but not the potential additional properties of the plant oils and waxes.
Previous studies have reported that regular oral intake of polyphenol-rich foods and supplements may protect the skin from sun damage, reduce the risk of chronic inflammatory diseases including arthritis, diabetes, and cancer, and improve recovery after exercise [20–23]. Potential benefits of topical phytochemical-rich plant extracts have been investigated in laboratory studies, but only in a limited number of clinical trials, despite their popular use in cosmetics [15, 24]. They have not previously been studied in the context of onycholysis. This study did not clarify which of the reported bioactive signaling pathways of the phytochemicals contributed the greatest benefit, but several possible explanations exist.
Its antioxidant properties may have potentially protected rapidly proliferating onychocytes from oxidative damage [12,13,15–17]. Gaultheria procumbens, particularly rich in methyl salicylate, may have reduced an inflammatory response against the nail bed tissue triggered by local chemotherapy damage, thus preventing further structural damage. These properties may also have reduced discomfort via local analgesic effects [11,14,25]. Tarchonanthus camphoratus, which contains phenolic acids, flavonoids, and tannins, also showed anti-inflammatory properties with a potency reportedly twice that of indomethacin. Furthermore, its bactericidal and fungicidal properties may have protected the nail bed from secondary infections, especially if splits or cracks had occurred [25,26]. Both Lavandula officinalis and Eucalyptus globulus contain lactones, flavonoids, tannins, phenolic acids, and terpene esters, which also exhibit anti-inflammatory, antifungal, and bactericidal properties [25,26]. The base waxes create a protective barrier for the skin, helping to retain moisture and thus preventing splitting and cracking of the nail bed, which makes it more difficult for pathogens to penetrate [24,26]. In contrast to standard petrolatum-based waxes, they also possess their own bioactive properties.
Olea europaea, predominantly oleic acid, has antioxidant and anti-inflammatory properties [24] and, in a randomized laboratory study, reduced epidermal levels of 8-hydroxydeoxyguanosine, a marker of DNA damage, after animals were exposed to excessive sunlight [15]. Theobroma cacao and Butyrospermum parkii, FDA-approved natural bases in cosmetics, possess antioxidant properties due to their vitamin E and vitamin A content, and even after processing, Cera alba exhibits antibacterial and antifungal properties [24, 26]. Although some properties overlap, combining different plant extracts, each containing different phytochemicals, has been suggested to produce synergistic protective effects [24–26].
In summary, the large difference between the control and intervention balms in this double-blind, randomized study strongly suggests that the phytochemicals in natural oils and waxes were sufficiently absorbed into the nail beds to protect participants from the harmful effects of chemotherapy through their bioactive properties. In this study, participants began using the balm at or near the start of chemotherapy; therefore, the study did not address whether this balm could prevent further deterioration or provide relief if damage had already occurred. Which properties and ingredients provided the most relevant benefit remained unanswered. Further research in this area is clearly warranted, as it could lead to improved versions of this and other topical therapies. Further research combining different versions of the balm, with or without nail cooling, is planned to evaluate whether this provides additional protection against the debilitating onycholysis.